 One atoms electrons are unsymmetrical, which creates a temporary dipole. This strong dependence is seen by the 1/r^6 portion of the potential energy of van der waal forces equation (Ep=a1a2/r6), where a represents the polarizability of an atom or molecule. A temporary attraction between two adjacent atoms. In this diagram there are two forces, which, when added together give a resultant force of 30 newtons (N). Substances that are polar experience dipole-dipole interactions. The strength of the interaction is also dependent upon distance between atoms or molecules. Intermolecular Forces All substances experience dispersion forces between their particles. This increases the polarizability of the element, which increases the strength of potential energy of van der waal interaction. The meaning of each of these forces listed in the table above will have to be thoroughly understood to be successful during this unit. 
For example, when one element has a greater amount of electrons than another number, there is more distortion of electrons that may occur. 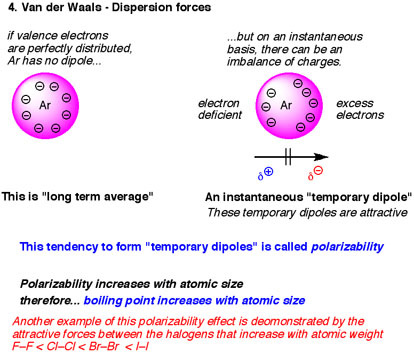
However, the strength of the potential energy of the the interaction varies by element, and thus the strength of the attractive force varies by element as well. These interactions are ALWAYS present and attractive, though they are weak. 
London/Dispersion forces, also known as Van Der Waal Forces or Induced Dipole-Induced Dipole, are weak, natural, electrostatic interactions between elements that are stimulating by fluctuating electron density, which in turn results in fluctuating dipoles. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
